Understanding Allograft vs Autograft vs Xenograft: A Complete Guide
Understanding Allograft vs Autograft vs Xenograft: A Complete Guide
Introduction to Transplantation Methods
When it comes to organ and tissue transplantation, medical professionals have several options at their disposal. Allograft, autograft, and xenograft represent the three main categories of transplantation methods used in modern medicine. Each approach offers unique advantages and challenges that must be carefully considered before proceeding with treatment.
Did you know that the first successful human organ transplant was performed in 1954? Since then, transplantation techniques have evolved tremendously, saving countless lives worldwide. However, understanding the differences between these procedures is crucial for both medical professionals and patients facing potential transplant situations.
In this comprehensive guide, we'll explore the fundamental differences between allograft, autograft, and xenograft procedures, examining their applications, success rates, rejection risks, and future developments in the field. Whether you're a medical student, healthcare professional, or simply curious about transplantation science, this article will provide valuable insights into this fascinating area of medicine.
What is an Allograft?
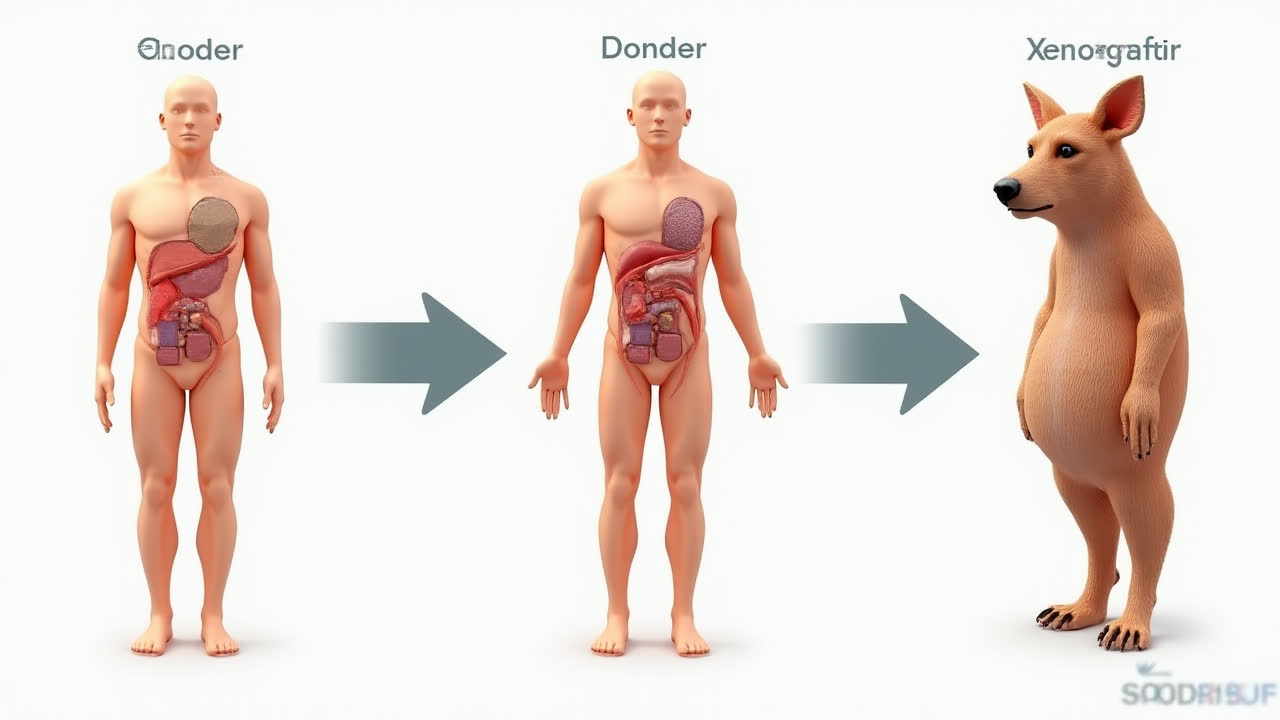
An allograft refers to tissue or organs transplanted from one person to another within the same species. The donor and recipient are genetically different individuals, though they must typically share compatible blood types for successful transplantation. Common allograft procedures include skin, cornea, heart, liver, kidney, and bone marrow transplants.
One of the most common sources for allografts is family members, as genetic similarities can improve compatibility and reduce rejection risks. However, non-related donors can also provide successful allografts when properly matched. The waiting list for organ donations demonstrates the significant need for allograft donors worldwide.
While allografts offer life-saving possibilities, they come with certain challenges. The primary concern is the risk of rejection, where the recipient's immune system recognizes the transplanted tissue as foreign and attacks it. To reduce this risk, recipients typically require immunosuppressive medications, which can leave them vulnerable to infections and other complications. I've seen patients struggle with finding the right balance between preventing rejection and maintaining a functioning immune system—it's often a delicate juggling act for both patients and their medical teams.
Allograft procedures have revolutionized treatments across various medical fields. In orthopedics, bone allografts help repair limbs and restore function. For severe burn victims, skin allografts provide crucial temporary coverage while their own skin regenerates. Corneal transplants have restored vision to countless individuals suffering from corneal damage or disease. The versatility of allografts makes them an invaluable resource in modern medicine.
What is an Autograft?
An autograft involves transplanting tissue from one part of a person's body to another location within the same individual. This approach eliminates rejection concerns since the tissue comes from the patient's own body. Commonly autografted materials include blood, bone, skin, and even specific proteins.
The beauty of autografts lies in their simplicity—they're essentially a form of self-donation. For example, during certain orthopedic procedures, surgeons might harvest a small piece of bone from the patient's hip, process it, and then use it to repair another bone in the body. Similarly, skin grafts for burn victims often utilize healthy skin from elsewhere on the patient's body.
Autografts offer exceptional reliability compared to other transplantation methods. Since the tissue is genetically identical to the recipient (being the same person), the chances of rejection are virtually non-existent. This eliminates the need for immunosuppressive drugs and their associated complications. Additionally, infection risks are significantly lower than with allografts or xenografts.
However, autografts do have limitations. The availability of tissue is restricted to what can be safely harvested from the patient. This creates obvious constraints in cases requiring large amounts of tissue or entire organs. Furthermore, the procedure creates two surgical sites—the donor site and the recipient site—potentially increasing recovery time and discomfort. Despite these challenges, autografts remain the gold standard when feasible due to their high success rates and minimal rejection risks.
What is a Xenograft?
A xenograft represents the most biologically distant transplantation approach, involving tissue or organs transferred from one species to another. The most common example in research is the transplantation of human tumor cells into immunocompromised mice for cancer studies. In clinical applications, xenografts may involve using animal tissues or organs in human recipients.
Xenotransplantation has been explored as a potential solution to the worldwide organ shortage crisis. With thousands of patients on waiting lists for life-saving organs, researchers have investigated using animal organs—particularly from pigs—as alternative sources. Pigs have become the primary focus due to their similar organ sizes and physiological compatibility with humans.
However, xenografts face significant challenges. The risk of rejection is extremely high due to the substantial genetic differences between species. The human immune system vigorously attacks xenografts, making long-term survival difficult without powerful immunosuppression. Additionally, concerns exist regarding the potential transmission of animal diseases to humans and the ethical implications of using animals as organ sources.
Recent advancements in genetic engineering have offered new hope for xenotransplantation. Scientists have created genetically modified pigs with human-like tissue characteristics, potentially reducing rejection risks. In January 2026, surgeons performed the first pig-to-human heart transplant, marking a significant milestone in xenograft research. Though the patient ultimately died two months later, the procedure demonstrated the potential viability of this approach and highlighted areas for further development.
Comparative Analysis: Allograft vs Autograft vs Xenograft
| Feature | Allograft | Autograft | Xenograft |
|---|---|---|---|
| Source | Another person (same species) | Same individual | Different species |
| Rejection Risk | Moderate to High | Minimal to None | Very High |
| Infection Risk | Moderate | Low | High |
| Reliability | Moderate | High | Low |
| Availability | Limited (donor dependent) | Limited (patient tissue dependent) | Potentially abundant |
| Immunosuppression Needed | Yes | No | Extensive |
| Common Applications | Skin, corneas, heart, liver, kidney, bone marrow | Skin, bone, blood vessels, fat | Research, heart valves, experimental organ transplants |
| Current Clinical Use | Widespread | Widespread | Limited/Experimental |
Clinical Applications and Future Developments
Each transplantation method serves specific clinical needs based on its unique characteristics. Autografts remain the preferred option whenever possible due to their minimal rejection risk and high success rates. However, they're limited by the amount of tissue available from the patient. I remember consulting with a burn specialist who explained that for patients with extensive burns covering most of their body, there simply isn't enough healthy skin available for autografting—making allografts or synthetic alternatives necessary.
Allografts have transformed numerous medical fields. Bone marrow transplants have revolutionized treatment for leukemia and other blood disorders. Corneal transplants restore vision to those with damaged corneas. Heart, liver, and kidney transplants save thousands of lives annually. The main limiting factor for allografts remains donor availability, with waiting lists for organs far exceeding supply in most countries.
Xenografts currently play a more limited clinical role but hold significant future potential. Certain xenograft products, such as pig heart valves treated to remove cells that could trigger rejection, have been used successfully for decades. More complex xenotransplantation, such as whole organs, remains experimental but is advancing rapidly with new genetic engineering techniques.
The future of transplantation likely involves a combination of approaches. Bioengineering advancements may soon allow scientists to grow organs from a patient's own cells, essentially creating custom autografts without donor site complications. Meanwhile, 3D bioprinting technologies are being developed to create tissue structures that could supplement or eventually replace traditional transplantation methods. These innovations, combined with improvements in existing techniques, promise to expand treatment options and address the critical shortage of transplantable tissues and organs.
Frequently Asked Questions
Which transplant type has the lowest rejection rate?
Autografts have the lowest rejection rate among all transplant types because they use the patient's own tissue. Since the transplanted material is genetically identical to the recipient, the immune system recognizes it as "self" rather than foreign material. This eliminates the need for immunosuppressive medications and their associated complications. However, autografts are limited by the amount of tissue that can be safely harvested from the patient.
Why are xenografts not commonly used in clinical practice?
Xenografts face several significant challenges that limit their widespread clinical use. The primary obstacle is the extremely high risk of rejection due to substantial genetic differences between species. Additionally, concerns exist about potential disease transmission from animals to humans (zoonosis), the accelerated aging of animal tissues compared to human tissues, and various ethical considerations. Despite these challenges, research continues with genetically modified animals that may eventually make xenotransplantation more viable for addressing organ shortages.
What factors determine which transplant method is used for a patient?
Multiple factors influence the selection of transplant method for a patient. These include the type and amount of tissue needed, the urgency of the procedure, the availability of donor tissue, the patient's overall health status, and the potential for rejection. Generally, autografts are preferred when feasible due to their minimal rejection risk. Allografts become necessary for entire organ transplants or when sufficient autograft tissue isn't available. Patient-specific factors such as age, immune system status, and ability to tolerate immunosuppressive medications also play crucial roles in determining the most appropriate transplantation approach.
Conclusion
Understanding the differences between allograft, autograft, and xenograft transplantation methods is essential for both medical professionals and patients navigating the complex world of tissue and organ replacement. Each approach offers distinct advantages and challenges that must be carefully weighed against individual patient needs and clinical circumstances.
Autografts, with their minimal rejection risk, remain the gold standard when feasible. Allografts provide life-saving options for many patients but require careful matching and immunosuppression management. Xenografts, while still largely experimental for complex applications, hold promise for addressing the critical shortage of transplantable human tissues and organs.
As medical science advances, we can expect continued refinement of existing transplantation techniques alongside revolutionary new approaches like bioengineering and 3D printing of tissues. These developments offer hope to millions worldwide awaiting transplants and may eventually transform our fundamental approach to tissue and organ replacement.